Modern materials and technologies guarantee a high degree of color reliability, reproducibility and efficiency in the manufacture of zirconium oxide dental prostheses. This makes it all the more annoying when, despite careful consideration of all important aspects of the laboratory process, discoloration suddenly occurs after sintering. A common reason for this is oxidic emissions from the heating elements and their deposits in the furnace chamber.
Especially with regard to highly esthetic monolithic zirconia restorations, it is important to understand and avoid this effect.
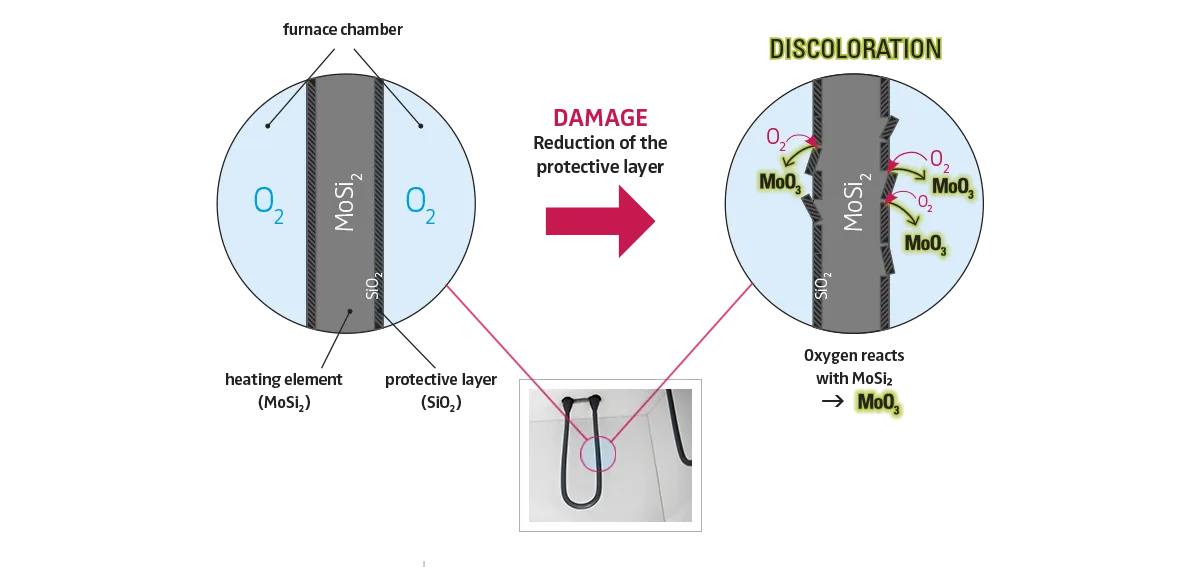
How is MoO3 produced?
Intact heating elements of MoSi2 are surrounded by a protective layer of SiO2 (silicon dioxide). This glass-like layer builds up on the heating element at high temperatures (oven-specific between 1000°C and 1600°C) and shields the underlying MoSi2.
However, the SiO2 layer is only under low compressive residual stress. If external stresses acting on the glass-like protective layer become too great, cracks and chipping will occur. If the MoSi2 is now exposed locally or over a large area, MoO3 is formed under the influence of oxygen in the furnace chamber during the subsequent heating phase (so-called 'pest oxidation', Fig. 2)
Fig. 3: Illustration of the development of discoloration caused by MoSi2 heating elements. The heating element consumes itself in the course of time
Stresses that lead to damage of the SiO2 layer can occur in different ways. In the course of time and after many sintering cycles, the SiO2 protective layer itself can grow so much that the residual compressive stresses are no longer sufficient and the layer becomes brittle. In a certain way, such defects are therefore part of the life cycle of MoSi2 heating elements.
However, damage can be drastically accelerated by the temperature control during sintering. In particular, very slow heating rates in the lower temperature range (oven-specific between 400°C and 600°C) or fast temperature changes in the high temperature range, such as those occurring during speed sintering, have a negative effect on the integrity of the SiO2 protective layer.
Tip:
If you include a small, white piece of zirconium oxide (e.g. from the edge of a used blank) for daily inspection, you will know when to clean at the latest.