... but not yet fixed dentures.

Article by Nadine Heilemann,
Product manager
It is likely that 3D printing will play an important role in the process chains of future digital dental technology and medicine. But where are we today and what is technically possible?
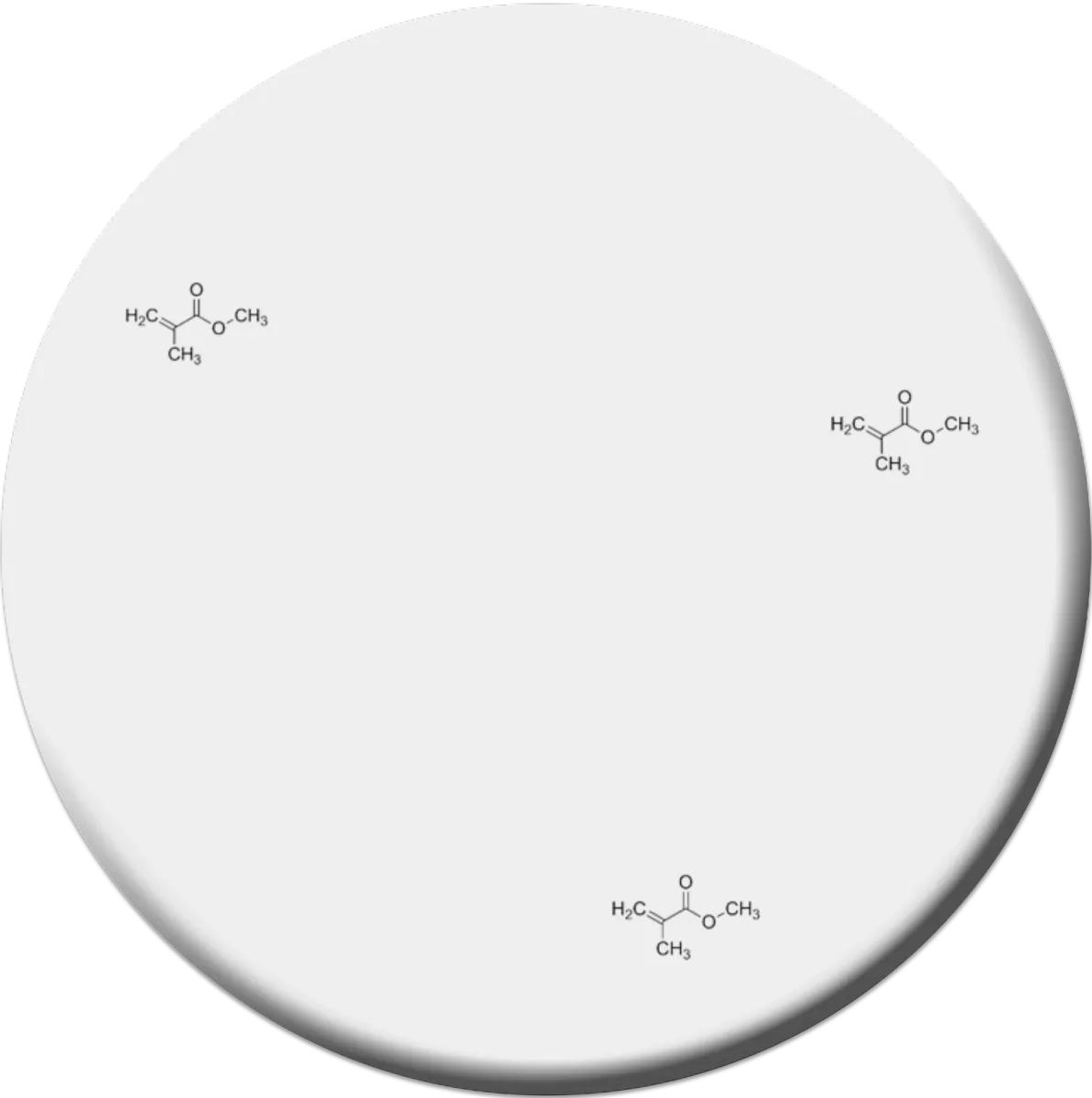
There are a number of dental indications that are interesting for additive manufacturing. However, the euphoria and willingness to invest that we saw three to four years ago seems to have cooled down a bit. There has been some disillusionment with printed, polymer-based medical devices. The oral cavity is a "hostile" environment and places high demands on denture materials. In addition to fitting well, good biocompatibility, mechanics and longevity are conditions that 3D printed dental structures must meet.
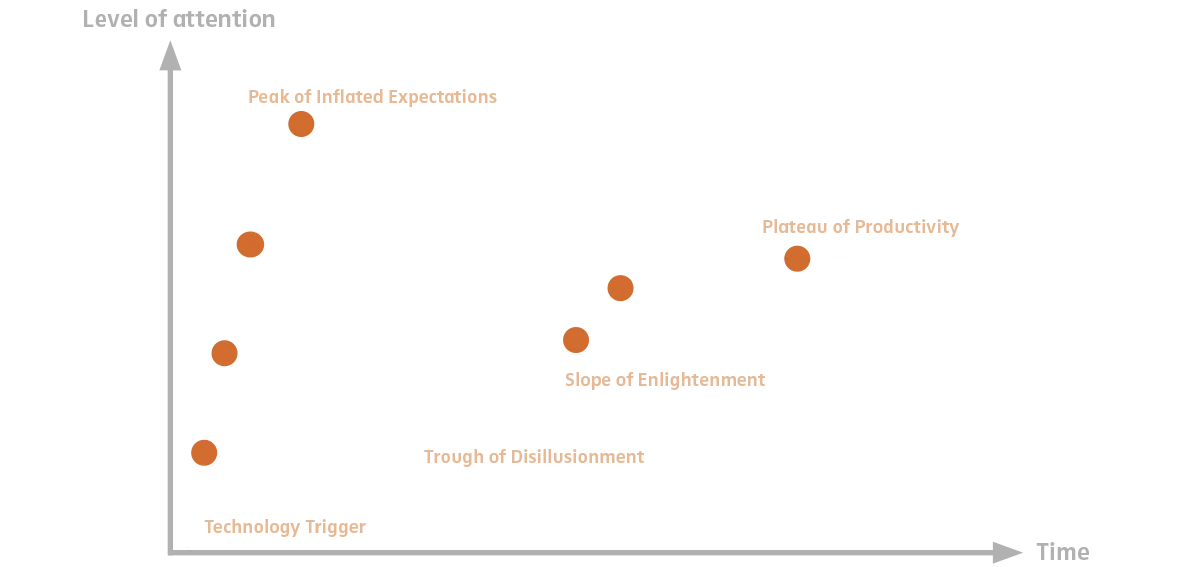
Explanation of the illustration:
The hype cycle represents the different phases of public awareness that new technology goes through when it is introduced. In terms of 3D printing, the following can be noted: The expectation that medical devices can be printed right out of the box that can compete with milled designs in terms of mechanics, biocompatibility and aesthetics is - for the time being - disappointing. However, 3D printing makes particular sense for the production of dental models, i.e. non-medical products.
Phase 1: „Technology Trigger”: The trigger is the introduction or announcement of new technology.
Phase 2: „Peak of Inflated Expectations”: The new technology generates a lot of attention and high expectations.
Phase 3: „Trough of Disillusionment”: Expectations are not met (printing of medical devices, e.g. bite splints), attention is waning.
Phase 4: „Slope of Enlightenment”: Despite decreased public attention, the technology is further developed and finds its application areas (non-medical devices, e.g. models).
Phase 5: „Plateau of Productivity”: The technology is recognized, establishes itself in the application areas and continues to mature.
Source: Edelhoff D., Güth J.-F., Trimpl J., Schweiger J., 3D-Druck in der Zahnheilkunde – zwischen Hype und Produktivitätssteigerung, in: BZB 9/2018, S. 46 - 55. The authors' assessment of the status of the individual applications based on a modification of the Gartner Hype Cycle with regard to the application of 3D printing in dentistry.